1. What isolation should the patient be
put in?
Patients should be put in enteric
isolation. Enteroviruses are transmitted mostly by the fecal-oral route, but
some serotypes may be spread by respiratory secretions or fomites.
2. What is known about the pathogenesis of the infection?
Differences in pathogenesis for the
enteroviruses mainly result from differences in tissue tropism and cytolytic
capacity of the virus.
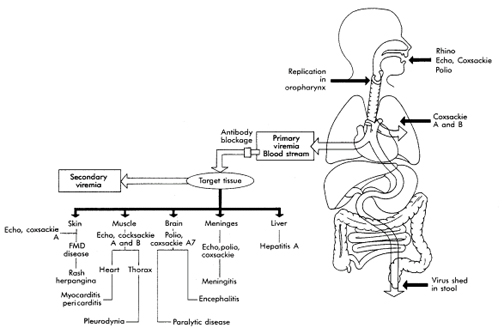
The upper respiratory tract, the
oropharynx, and the intestinal tract are the portals of entry for the
enteroviruses. The virions are impervious to stomach acid, proteases, and
bile. The virus initiates replication in the mucosa and lymphoid tissue of
the tonsils and pharynx and later infects lymphoid cells of Peyer's patches
underlying the intestinal mucosa. Primary viremia spreads the virus
to receptor-bearing target tissues, where a second phase of viral
replication may occur, resulting in symptoms and a secondary viremia. Virus
shedding from the oropharynx can be detected for a short time before
symptoms begin, whereas virus production and shedding from the intestine may
last for 30 days or longer, even in the presence of a humoral immune
response.
Coxsackieviruses and echoviruses recognize
receptors expressed on many cell types and tissues and cause a broad
repertoire of disease. These and other enteroviruses recognize receptors on
cells of the central nervous system, heart, lung, pancreas, and other
tissues.
Most enteroviruses are cytolytic,
replicating rapidly and causing direct damage to the target cell.
Antibody is the major protective immune
response to the enteroviruses. Secretory antibody can prevent the initial
establishment of infection in the oropharynx and gastrointestinal tract, and
serum antibody prevents viremic spread to the target tissue and therefore
disease. Serum antibody is generally observed 7 to 10 days after infection.

Figure 1. Pathogenesis of enteroviruses. The target tissue infected by the enterovirus determines the predominant disease caused by the virus.
3. How is the viral infection detected?
The finding of >5 white blood cells/mm3
of CSF is abnormal and is indicative of meningitis. The patient was treated
for bacterial meningitis because of the predominance of neutrophils in the
CSF. Early in the course of viral meningitis (the first 24 to 48 h),
neutrophils can be predominant. However, mononuclear cells are predominant
later in the disease. The normal CSF glucose level also argues against
bacterial meningitis. Finally, a positive CSF viral culture confirms the
diagnosis of viral meningitis.
4. Why was there a neutrophil predominance on the LP?
Neutrophil is a common finding in early viral meningitis, but as indicated above, mononuclear cells predominate later in the disease.
5. What is the appropriate treatment for
this viral infection?
The management of patients with
enteroviral infections includes supportive care. Infection control measures
are recommended for patients and their families to interrupt transmission of
virus to others who may be susceptible. These include handwashing. This is
of particular importance in day-care centers, where agents that are spread
by the fecal-oral route are of particular concern.
A new drug that inhibits the uncoating of
picornavirus particles is being evaluated in clinical trials. Pleconaril
binds reversibly to a hydrophobic pocket in the outer coat protein (VPI) of
picornaviruses. Binding in this site prevents uncoating of the virion RNA
following attachment and penetration of the cell membrane. Inhibiting this
step prevents replication of the RNA and production of progeny virions. The
compound has undergone extensive in vitro sensitivity testing against over
215 clinical isolates of enterovirus strains known to cause serious human
disease; over 90% of the 100 known rhinovirus serotypes at a concentration
of 1 ug/ml. Pleconaril was protective in several mortality studies conducted
in weanling, suckling and adult mice infected with several different strains
of coxsackievirus.
Clinical Studies
Pleconaril has been studied in over 700
human adult and pediatric volunteers and patients in 17 completed clinical
studies to date, as well as in six ongoing placebo controlled trials of the
clinical trials conducted to date, pleconaril has been very well tolerated,
with an aggregate side effect profile similar to placebo.
Phase I
Pleconaril exhibits dose-dependent linear
pharmacokinetics resulting in plasma levels at pharmacodynamic steady state
that are in excess of levels required to inhibit viral replication of
rhinoviruses and enteroviruses. Pediatric pharmacokinetics are similar to
that of adults, requiring no dosage adjustment for children. Neonatal
pharmacokinetic studies are currently underway.
Viral Meningitis
In a placebo controlled adult enteroviral
meningitis study, pleconaril treatment resulted in a statistically
significant shortening of disease duration (54% reduction, 11 vs. 5 days; p
= 0.0005) the primary endpoint of the study. Elements of the primary
endpoint included clinically important symptoms of viral meningitis such as
a reduction in severe incapacitating headache, which usually leads to
hospitalization. Pleconaril was statistically superior to placebo in the
secondary endpoints of the time to complete absence of headache (50%
reduction, 14 vs. 7 days; p = 0.014), duration of analgesic use (50%
reduction, 12 vs. 6 days; p = trends favoring pleconaril were also observed
in other secondary endpoints, including a reduction in the time to return to
work and a reduction in the time return to full levels of leisure activity.
Viral Respiratory Infections
Pleconaril demonstrated pronounced and
clinically significant reductions in respiratory and systemic symptoms
resulting from the intranasal administration of a safety-tested strain of
coxsackievirus A-21 in health volunteers when compared to placebo. Clinical
studies are currently underway in vial respiratory disease in odds media
prone children, patients with asthma, and otherwise normal subjects.
Pharmaceutics
Pleconaril is supplied as a
cherry-peppermint flavored oral liquid containing pleconaril at a
concentration of 40 milligrams per milliliter in medium chain triglycerides
(Miglyol 810) 2 % surfactants (Tween 80 and Arlacel) as well as 0.15%
saccharin. This formulation is palatable when taken by children and adults.
A tablet dosage form has been developed for the adult market.
6. Would this infection be likely to occur
in February?
No. Enterovirus meningitis outbreaks
generally occur in the late summer and early fall.